
Redefining Enzyme Catalysis: Chemical Control in the Biosynthesis of Terpenes
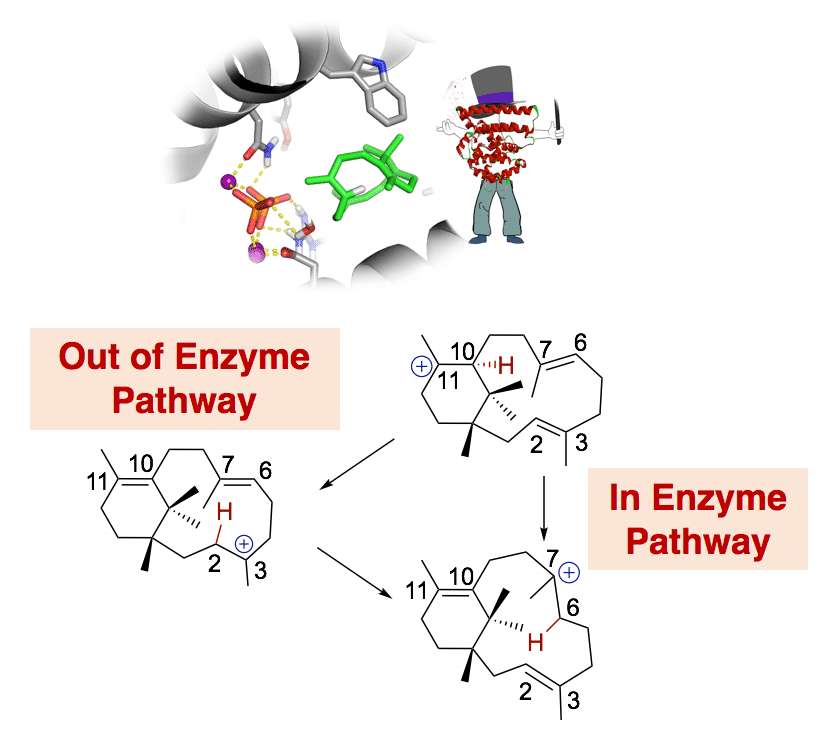
Terpene cyclases are responsible for the initial cyclization cascade in the multistep synthesis of more than 80,000 known natural products. This abundance of compounds is generated using a very limited pool of substrates based on linear isoprenoids. The astounding chemodiversity obtained by terpene cyclases suggests a tremendous catalytic challenge to these often-promiscuous enzymes. In the current presentation, we will focus on representative mono-, sesqui-, and diterpene cyclases. Specifically, we will describe a novel view of enzyme catalysis based on the biosynthesis of these terpene synthases. For these enzymes, the catalytic challenge is chemical control of hyper-reactive intermediate carbocation species, rather than rate acceleration. Based on multiscale simulations, we propose that to ensure catalytic specificity these enzyme wizards employ their active site contour to fold the initial substrate and sequester intermediates. Chemical control is obtained through conformational selection, electrostatic steering, dispersive interactions that adapt to changing electronic requirements during catalysis, as well as carefully placed basic residues. Nonetheless, this catalytic toolbox is not sufficient for exclusive product formation, providing a rationale for fidelity in the largely promiscuous terpene cyclases.1-4
- Weitman, M.; Major, D. T. Challenges posed to bornyl diphosphate synthase: Diverging reaction mechanisms in monoterpenes. J. Am. Chem. Soc. 2010, 132, 6349-6360.
- Major, D. T.; Weitmann, M. Electrostatically Guided Dynamics - the Root of Fidelity in a Promiscuous Terpene Synthase? J. Am. Chem. Soc. 2012, 314, 19454-19462.
- Major, D. T.; Freud, Y.; Weitman, M. Catalytic Control in Terpenoid Cyclases: Multiscale Modeling of Thermodynamic, Kinetic, and Dynamic Effects. Curr. Opin. Chem. Biol. 2014, 21, 25-33.
- Dixit, M.; Weitman, M.; Gao, J.; Major, D. T. Chemical Control in the Battle Against Fidelity in Promiscuous Natural Product Biosynthesis: The Case of Trichodiene Synthase. ACS Catalysis 2017, 7, 812-818.
Powered by Eventact EMS