First-in-Human Transcatheter Tricuspid Valve Repair by the Mistral Device
[Mitralix Ltd.]
Background: Mistral is catheter-based treatment for tricuspid (TR) and mitral (MR) regurgitation. This single use system consists of a 8.5F delivery system carrying a spiral-shape device to be implanted in the right ventricle (TR) or left ventricle (MR). The Mistral mode of action: improve leaflets coaptation by gently grasping chords from two leaflets bringing them together; hence alleviating symptoms and improving valve and ventricular function. Four compassionate and four FIM study cases were performed previously showing procedural safety, device stability and encouraging clinical improvement. This report of the latest case performed in the FIM trial.
|
Before rotations |
After rotations |
Mistral |
Procedure and results:
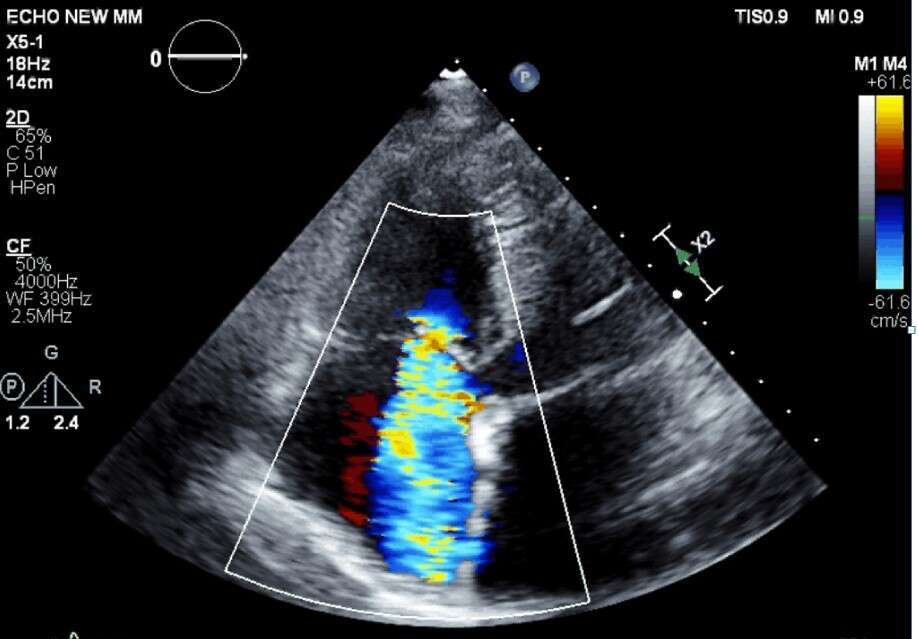
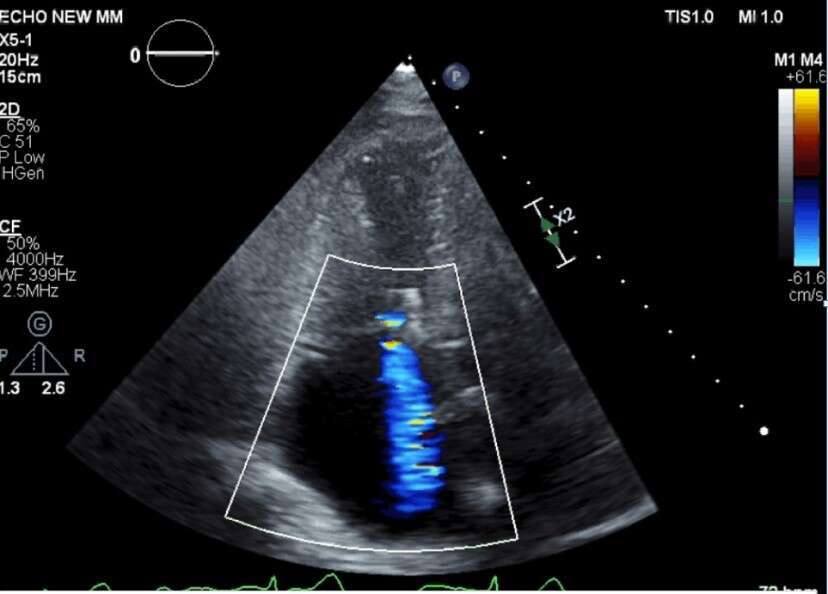
Female patient (73 years old) suffering from severe TR with severe right sided heart failure, was declined of surgery (high surgical risk). Procedure was performed under general anesthesia with continuous TEE monitoring and guidance. Mistral implanted, grasping chordae of tricuspid anterior and septal leaflets. Procedural duration 75 minutes. Successful grasping was obtained. Patient was hemodynamically stable throughout and following procedure, no adverse events were observed, patient was discharged one day post procedure. TR grade was clearly improved as observed during procedure by TEE and comparing screening and discharge TTE. Marked improvement and recovery of right ventricular function was recorded. Full echocardiographic and clinical will be presented.
|
Screening TR EROA - Severe |
Discharge TR EROA - Mild |
Conclusions:
The Mistral provides an innovative transcatheter approach for treating tricuspid valve insufficiency. Our initial experience provides a proof of concept and demonstrates safety and short term efficacy of this technology.
Powered by Eventact EMS
 before rotation - Copy 3.jpg)
 after rotation - Copy 3.jpg)