
Flash
Perthio-bambusurils: Conformational unusual cavitands
2Schulich Faculty of Chemistry, Technion – Israel Institute of Technology, Haifa, Israel
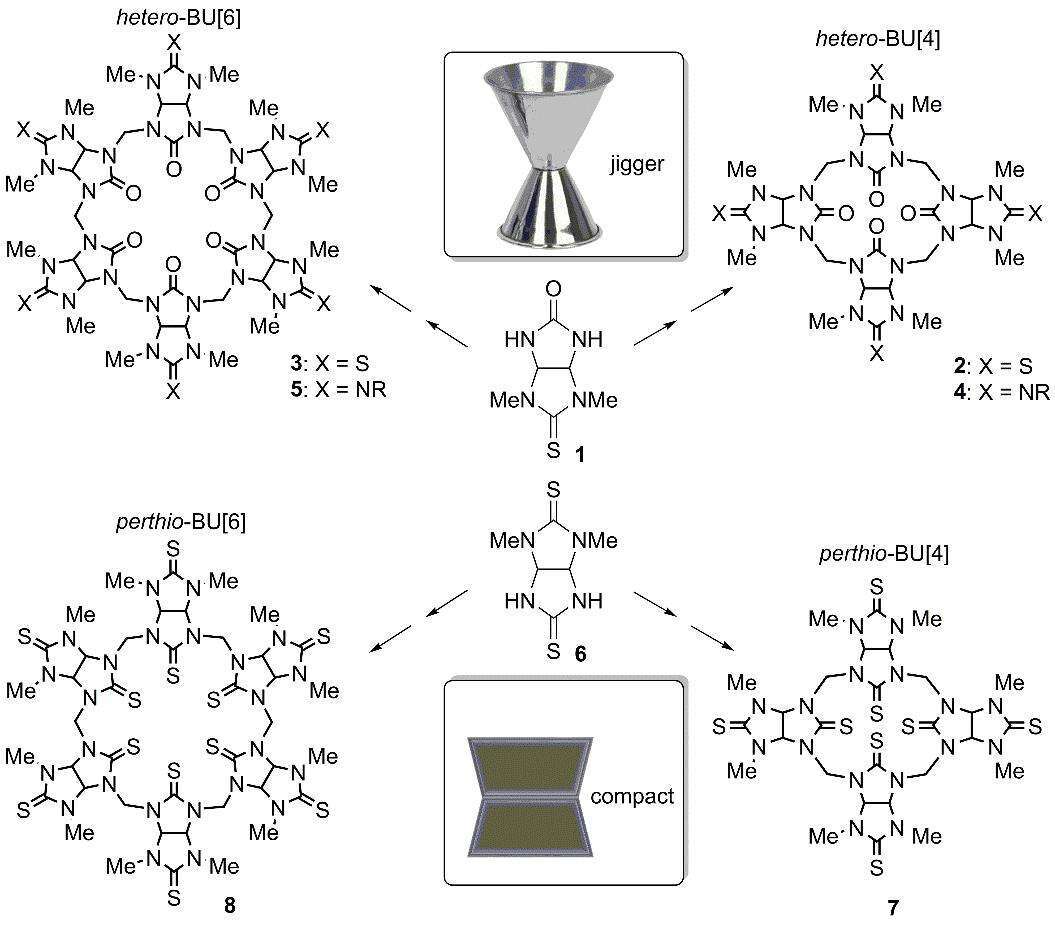
Heteroatom replacement in large molecules, such as biological macromolecules, may lead to significant conformational changes because each atom forms multiple intramolecular interactions that operate in concert. In contrast, heteroatom replacement in small molecules, including synthetic cavitands, although resulted in some changes in binding properties, such as coordination to metal ions, no significant change in the original conformation of the parent systems were observed. Similar conservation of the molecular conformation has been demonstrated in hetero-bambusurils (BUs).1 These anion binders are comprised of glycoluril or hetero-glycoluril units connected by methylene bridges, and are quite rigid cavitands due to restricted rotation around the single bonds. In all BUs the glycoluril fragments are practically frozen in an alternate arrangement, featuring an extended conformation that is reminiscent of a jigger, namely, two back-to-back cups sharing the same base. All BUs reported to date with equatorial oxygen atoms, including the parent BU[4]s and BU[6]s, semithio-BUs and semiaza-BUs exhibit the same jigger conformation.
Here we report the synthesis of perthio-bambusurils and demonstrate its unique yet unknown compact conformation, which has never been observed in the BU landscape, explaining their weak anion binding properties. These unusual observations originate from strong intramolecular attractive interactions between the equatorial sulfur atoms and multiple hydrogen atoms within the molecule Nevertheless, these novel macrocyclic cavitands bind thiophilic metal cations at their sulfur-edged portals.
[1]. Reany, O.; Mohite, A.; Keinan, E. Isr. J. Chem. 2018, 58, 449-460.
Powered by Eventact EMS