
Macrocyclic oligofurans: A computational study
Macrocyclic structures with a π-conjugated backbone often display unique structures, properties and functions. In many cases, the physical properties of linear oligomers are influenced by undesired chain-end effects. In this respect, corresponding fully π-conjugated macrocycles represent model systems that combine the ‘infinite’ defect-free π-conjugated chain of an idealized polymer with the advantage of a structurally well-defined oligomer, while excluding perturbing end-effects. This renders them interesting candidates for various future applications in organic and molecular electronics and for the study of host–guest interactions, aggregation, and self-assembly on surfaces.[1] While macrocyclic oligothiophenes (nCT) were extensively explored – both computationally and experimentally – macrocyclic oligofurans (nCF) were only recently synthesized in our group. [2] Following the recent introduction of linear long oligofurans,[3] we were interested in exploring the properties of macrocyclic oligofurans.
Here we report the computational study of macrocyclic oligofurans in syn- and anti-conformations at the B3LYP/6-311G(d) level of theory.[4] Strain energies, bond length alternations, HOMO−LUMO gaps, ionization potentials, reorganization energies, and NICS values are among the properties studied. Our results indicate that contrast to nCF, where small macrocycles display non-planar conformations, nCF with 6-8 units display relative planarity (Figure) with a strong quinoid character and low HOMO–LUMO gaps. We therefore conclude that 6CF-8CF should be ideal the ideal candidates to perform as organic semiconductors.
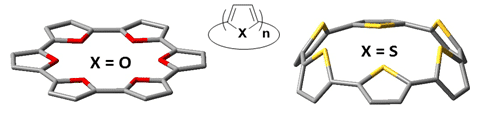
Figure. Calculated structures (DFT/B3LYP/6-311G(d)) of syn- 6CF (left) and 6CT (right).
[1] (a) M. Iyoda, H. Shimizu, Chem. Soc. Rev. 2015, 44, 6411-6424; (b) P. Bauerle, Angew. Chem. Int. Ed. 2000, 39, 3481-3486
[2] S.V. Mulay, O. Dishi, O. Gidron, in preperation.
[3] O. Gidron, M. Bendikov, Angew. Chem. Int. Ed. 2014, 53, 2546-2555
[4] O. Dishi, O. Gidron, J. Org. Chem., 2018, 83 (6), pp 3119–3125
Powered by Eventact EMS