DETECTION OF ALPHA-SYNUCLEIN AGGREGATES IN SKIN BIOPSIES OF PARKINSON’S DISEASE PATIENTS USING SUPER RESOLUTION MICROSCOPY
2Computer Science and Life Science, Open University of Israel, Ra'anana, Israel
3Specialty Research and Development, Teva Pharmaceutical Industries, Ltd., Netanya, Israel
4Neurology, Meir medical center, Kfar Saba, Israel
5Neurology, Tel Aviv Sourasky Medical Center, Tel Aviv, Israel
6Sagol School of Neuroscience, Tel Aviv University, Tel Aviv, Israel
7Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv, Israel
α-synuclein (α-Syn) aggregates in the central nervous system (CNS) have been associated with the development of Parkinson’s Disease (PD) and are currently its main pathological hallmark. α-Syn aggregation has also been identified in many peripheral tissues such as skin, suggesting a novel and accessible method for the detection of the disease.
The main goal of this research was to establish α-Syn in skin biopsies as a comparative biomarker for the detection of PD pathology by utilizing super-resolution microscopy (SRM). This unique method, specifically the Direct Stochastic Optical Reconstruction Microscopy (dSTORM) that we applied, provides the opportunity to investigate the α-Syn aggregation composition at the single-molecule level, and perhaps might overcome the challenge to identify small (sub-diffraction) α-Syn aggregates already in early stages of their formation. We have recently quantified and characterized nano-sized α-Syn aggregates in brain slices from a transgenic mouse model and showed correlation between aggregates’ size and disease severity and reversal by a specific drug, Anle138b. These α-Syn aggregates couldn’t be detected using conventional microscopy, thus demonstrating the distinct advantage of SRM in detecting small α-Syn aggregates and provides proof of concept for the current study.
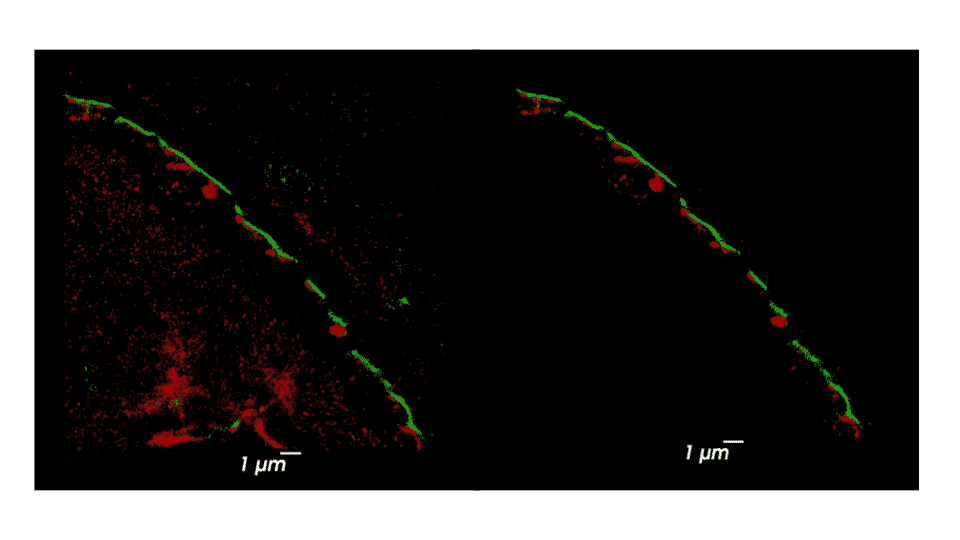
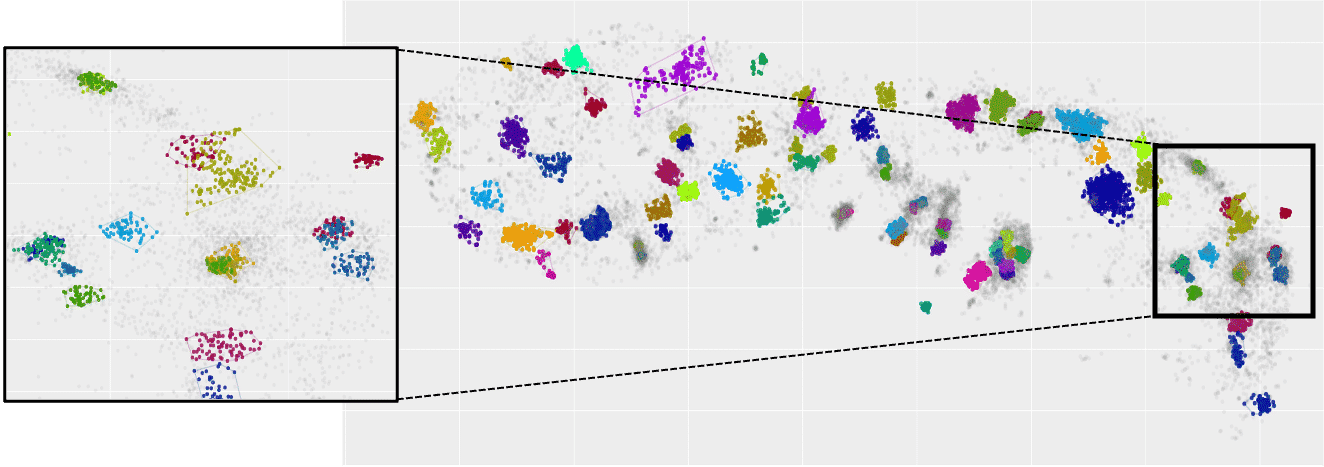
Using SRM, we imaged phosphorylated and total α-Syn in immunostained sweat glands of skin biopsies from volunteering PD patients and control subjects, who were recruited and clinically assessed at Tel-Aviv Sourasky Medical Center, Meir Hospital and Sheba Medical Center. Areas where colocalization of α-Syn and sweat glands’ membranal innervation is found are selected as regions of interest for further analysis. We applied a series of cluster analysis algorithms (DBSCAN, HDBSCAN & FOCAL) on the dSTORM output to characterize the precise size, density, and composition of α-Syn aggregates and their distribution within the sweat glands’ innervation, in order to explore possible correlations between the structural classifications and presence of the disease. Each cluster-identification algorithm can be adjusted to find slightly different classes of aggregates differing in size, density, and shape. Such adjustments in the algorithms’ parameters allow for classification of a range of aggregates, from small and dense to large and sparse. We will describe preliminary results and the characterization of α-Syn aggregation in skin biopsies. This novel method may establish sub-diffraction α-Syn aggregates as a new biomarker for PD and may be used to explore early changes in the prodromal stage of PD, by identifying individuals at risk years before PD onset. Furthermore, it might allow adjusting the most compatible treatment for each patient, according to the specific characterization of their pathological α-Syn aggregates manifestation.